We do not diagnose disease or recommend a dietary supplement for the treatment of disease. You should share this information with your physician who can determine what nutrition, disease and injury treatment regimen is best for you. You can search this site or the web for topics of interest that I may have written (use Dr Simone and topic).
“We provide truthful information without emotion or influence from the medical establishment, pharmaceutical industry, national organizations, special interest groups or government agencies.” Charles B Simone, M.MS., M.D.
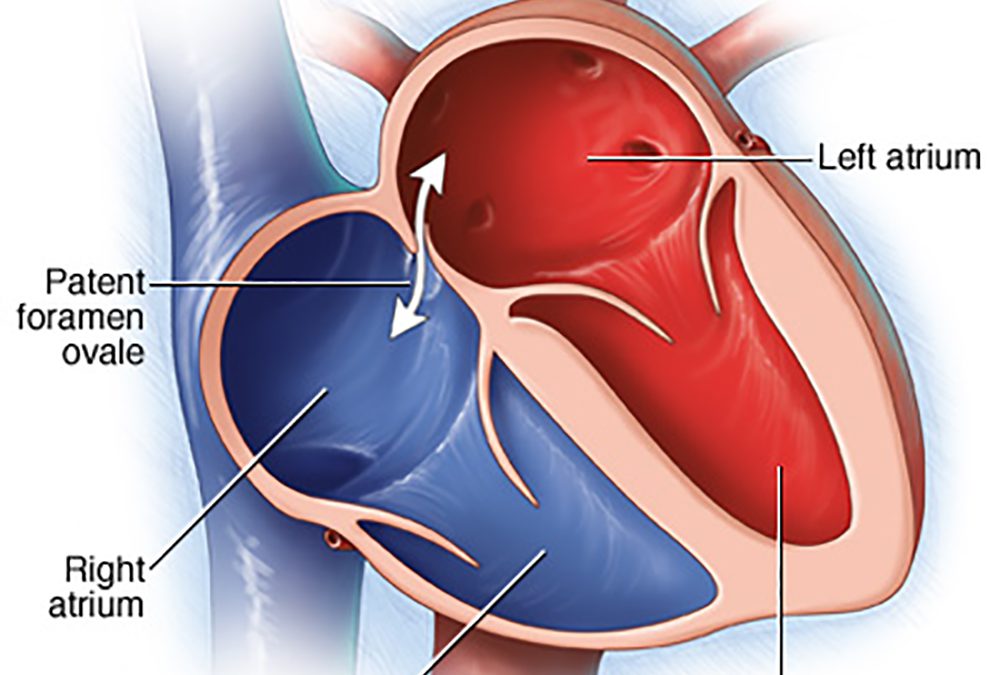
40% HAVE A PFO (PATENT FORAMEN OVALE) – DO YOU?
Lawrenceville, NJ (Dr Simone) – Do you get Jet Lag, fatigue, shortness of breath, chest pain, confusion, headaches, rapid heartbeat, or feel overheated, or have had an unexplained stroke, heart attack, or partial blindness? PFO (PATENT FORAMEN OVALE) affects up to 40% of all adults in the world and was first described by Claudius Galen in 200 A.D. It’s a hole in the upper chambers of the heart that usually closes during early infancy. Normally the blood travels from the right side of the heart through the lungs to the left side of the heart and then into the rest of the body before returning back to the right side of the heart. This allows the lungs to pick up oxygen and get rid of carbon dioxide (gas exchange), filter clots/particles out of the circulation, and cool the blood, and, therefore the body.
However, people with a PFO have an opening between the right and left upper heart (atria) that can be small in some people and larger in others. No matter what the size of the hole while resting, it becomes stretched and larger when the person sits or stands from a lying position, lifts something heavy, or bears down, as if having a bowel movement.
The incidence of PFO decreases with age, declining from 34% in people between the ages of 0 and 29 years to 25% in people between the ages of 30 and 79 years to 20% in people between the ages of 80 and 99 (1). However, the average size of the PFO increases with age from 3.4 mm during the first 10 years of life to 5.8 mm during the 10th decade of life (1). Even though up to 40% of the adult population have a PFO, little research has examined its impact on normal physiological processes.
At the end of a normal inspiration during diastole, the PFO allows a potentially significant amount of blood to bypass the lungs especially during exercise, at high altitude, and in patients with pulmonary hypertension. This does not occur with every heart beat. That portion of the blood that does not go through the lungs will:
1) not pick up oxygen and get rid of carbon dioxide causing problems at rest and high altitude;
2) not be filtered of blood clots or air bubbles. Consequently, unfiltered clots and air bubbles can go directly to the brain (STROKE), heart arteries (HEART ATTACK), eye (BLINDNESS), migraines with aura, kidney, any other organ;
3) not get cooled, resulting in a higher core temperature that results in an even lower oxygen level.
For many years only the issue of unfiltered blood clots causing a stroke was used to decide how to treat a patient with a PFO because the other important physiological consequences were not considered: including hypoxia, carbon dioxide retention, higher core temperature, and unfiltered air bubbles.
THE LARGER THE HOLE, THE WORSE THE PROBLEMS
REDUCED GAS EXCHANGE EFFICIENCY – less oxygen acquired and more carbon dioxide retained for the person with a PFO for two reasons: 1) simply less blood flow to the lungs; 2) gas exchange is less efficient (2). The size of the hole matters and not just having a PFO. The larger the hole, the less oxygen is picked up. Older patients (>65) with PFOs who also have heart failure with or without pulmonary hypertension have lower blood oxygen levels (SaO2) at rest (3). Surgical closure of the PFO improves arterial oxygenation at rest and during exercise, thus showing that blood flow through a PFO definitely has a measurable impact on gas exchange (4).
% OXYGEN SATURATION ( SaO2%) refers to the amount of oxygen picked up by red blood cell’s hemoglobin in the lungs. It is measured by a device called a pulse oximeter (pulse ox) put on a finger. The normal range is 97% to 100%. Below 95% is considered low for people without diseases and oxygen is usually administered if the level is about 92% or below. The normal range for a person with severe Chronic Obstructive Pulmonary Disease is 88%-92%.
CONSEQUENCES OF HYPOXIA – LOW BLOOD OXYGEN
1). Shortness of breath, fatigue, chest pain, confusion, headache, rapid heartbeat that results in less volume of blood pumped by the heart (decreased stroke volume). Blood levels below 80% can compromise the brain and heart. Continued low oxygen levels may lead to respiratory or cardiac arrest and loss of consciousness.
2). Constriction of small pulmonary arteries and dilatation of systemic arteries
3). Hypercoagulable state (increased by two-fold to eight-fold) causing clots is caused by acute low-pressure hypoxia at high altitude (5).
4). Higher rate of death and morbidity for outpatients with pneumonia when their oxygen saturation is less than 90% (6).
5). Jet Lag is caused by this fall in oxygen saturation and altitude sickness.
BODY TEMPERATURE
When the core temperature rises in a person without a PFO, the body responds by taking deeper breaths more rapidly to cool down the blood in order to maintain a constant brain temperature.
PFO patients have a higher core temperature at rest and during exercise (about 0.4 degrees C) because some of the blood does not go through the lungs to get cooled. This higher temperature results in an even lower oxygen level (because of a right-shifted oxyhemoglobin dissociation curve). The SaO2% is reduced by 1.5% – a significant amount (2). Therefore, on a hot day in the sun, these patients can become fatigued and lethargic within minutes due to the higher core temperature and lower oxygen level without any significant blood pressure reduction. They have a higher risk for heat exhaustion and heat stroke. These patients feel better when they put at least their feet in a cooler body of water – pool, lake, ocean, even cool water in a bathtub. Air conditioning may help, but some have had difficulty cooling down when given cool air to breathe.
HUMIDITY
Adding humidity to the air DECREASES the oxygen molecules because the water molecules displace oxygen and the other gases. The total pressure of air at sea level is 760 mm Hg (mercury) with or without humidity. Nitrogen is the most abundant gas comprising 79% of air’s concentration, followed by oxygen at 20.93% and carbon dioxide at 0.03%.
The concentration of oxygen in DRY air is calculated by multiplying 20.93% times 760 mm Hg = 159 mm Hg. This is the concentration of oxygen that enters the mouth and nose. As the air descends into the lungs where it gets humidified the concentration of oxygen decreases even more. At the level of the trachea of the lungs it is 149 mm Hg. At the alveoli, where gas exchange actually occurs, the oxygen concentration is 105 mm Hg because of the higher humidity at that level. When the atmospheric air is HUMID, there is even less oxygen in it and therefore less oxygen that enters the mouth and nose. As that air descends to the alveoli, the oxygen content is much lower compared to the DRY air example.
A person with a PFO will have a lower blood oxygen saturation in humid weather. If it is hot and humid, the person with a PFO has a much harder time breathing and fulfilling the body’s needs of oxygen.
HIGH ALTITUDE reduces breathing response, decreases the ability to oxygenate the blood and increases the susceptibility to Acute Mountain Sickness. A study (7) done on 21 healthy 20 year old subjects (11 with relatively small PFO) at a research facility at Mt. Chacaltaya, Bolivia, 5,260 m = 17,257 feet showed that gas exchange got worse for all people with or without a PFO when they were taken directly up to this high altitude. The oxygen saturation was about the same for those without or with a PFO: 67% vs 66%, respectively. For people without a PFO the oxygen saturation improved to 76% after 16 days of acclimatization because more oxygen is taken in by the lungs (increased alveolar ventilation).
But people with a PFO did not attain acclimatization after 16 days – both at rest or while exercising – and were still suffering from Acute Mountain Sickness (oxygen saturation = 70%). Symptoms of Acute Mountain Sickness include headache, anorexia, insomnia, breathlessness, and unsteady gait. They also had a higher risk for high altitude pulmonary edema (fluid in lung), stroke, heart attack, and damage to other organs.
Deep Breathing: One way to partially compensate for a decreased intake of oxygen at high altitudes is to slow down your breathing rate but increase the depth of your breaths. Instead of breathing quick and shallow and only in your lungs, breathe slowly and deeply and inhale until your stomach expands.
People who are born and live at high altitude have the same prevalence and sizes of PFOs as those who are low-altitude natives. Selective adaptation might have eliminated PFOs for those living at high altitude – but it did not. Tibetans, who thrive at altitudes over 4400 meters (14,435 feet) and whose ancestors have lived at high altitudes for thousands of years, have two gene variants (EGLN1 and PPARA) that help them use oxygen more efficiently with less hemoglobin than people who live at low altitudes – a process that may involve nitric oxide (8).
The following lists the ALTITUDE and % OXYGEN SATURATION for people WITHOUT a PFO.
0 feet 0 meters 97%
5,000 ft 1525 m 94%
6,000 ft 1825 m 93% Boeing 787 cabin altitude
8,000 ft 2440 m 91% Boeing 737 cabin altitude
8,000-10,000 ft 2440-3050 m 91% – 88% Learjet cabin altitude
10,000 ft 3050 m 88%
12,000 ft 3660 m 84%
13,120 ft 4000 m 80%
17,257 ft 5260 m 67% Mt. Chacaltaya, Bolivia
19,700 ft 6000 m 65%
26,250 ft 8000 m <50% DEATH ZONE
The % OXYGEN SATURATION for people WITH a PFO is lower.
People with a PFO traveling in a Learjet (cabin altitude 8,000-10,000 feet), Boeing 737 (cabin altitude 8,000 feet), or a Boeing 787 (cabin altitude 6,000 feet), will have an % OXYGEN SATURATION lower than 88%-91%, 91%, 93% respectively, and below 95% is considered low.
AIR FLIGHT
US Federal Aviation Regulations state that pressurized cabins must provide a cabin altitude of not more than 8000 feet at the maximum operating altitude of the airplane (FAA, 1996), so that the partial pressure of oxygen be maintained at 0.17–0.15 at sea level. Cabin altitude measurements by 28 airlines range from sea level to 8,915 feet.
The Boeing 737’s cabin altitude can climb to 8,000 feet while the aircraft’s actual altitude is 41,000. Newer aircraft such as the Boeing 787 are designed to have lower cabin altitudes of 6,000 feet. At 35,000 feet in a Learjet, the cabin altitude may be 8,000 to a maximum of 10,000 feet. There is less oxygen in the air at higher altitudes and our ability of get enough oxygen decreases. Alveolar oxygen tension decreases to 65 mm Hg at 8000 feet that reduces arterial oxygen tension to 60 mm Hg in healthy individuals (9). Patients with chronic hypoxia may have a greater reduction in SaO2 when they reach high altitude than those with healthy lungs. Any small reduction in the oxygen content of the air in the cabin results in a significant decrease in the SaO2 because of the oxyhemoglobin dissociation curve. This is even worse for those with PFOs.
The cabin pressure is mechanically increased by routing compressed air (“bleed air”) from the turbine engines to the air conditioning system. The cabin pressurization and ventilation is controlled by opening and closing of outflow valves via the pressure controller switch. This system pumps air into the aircraft that is adequately sealed to limit the rate at which this air escapes from the airplane cabin. Cabin altitude is a better physiological indicator than cabin pressure.
People are most comfortable when the plane climbs or descends at a rate of 500 feet per minute or less. Faster rates are uncomfortable and can cause painfully blocked ears and sinuses, and, in extreme cases, incapacitation. A turbine aircraft typically climbs and descends several thousand feet per minute. The pressure controller regulates the cabin altitude and varies the pressure at rates much lower than the aircraft’s actual altitude. For example, the cabin altitude climbs to 8,000 feet (taking 16 minutes at 500 feet per minute) while the aircraft actual altitude climbs to about 40,000 feet (at an average 2,500 feet per minute in the same 16 minutes).
There are two types of pressure control systems. The completely automated one requires the pilot to enter only the destination field elevation and then it schedules the timing and rate of cabin climb or descent. Older systems require the pilot to set the rate of a cabin climb/descent. After takeoff, the controller is set according to cruise altitude, and prior to descent it is adjusted to a level appropriate to the landing field elevation. This setting is usually 500 to 1,000 feet above the airport elevation.
If sudden cabin depressurization occurs in an aircraft like a Learjet flying at 35,000 feet, the pilots and passengers may initially experience a brief euphoria and then have as little as 5 to 12 seconds of useful consciousness to put on their oxygen masks. Following this brief period, without supplemental oxygen, their cognitive and motor skills diminish, leading to incapacitation, often with fatal consequences.
At 35,000 feet in a Learjet, the cabin altitude may be 8,000 to a maximum of 10,000 feet. If the cabin altitude rises above 10,000 feet, a device developed by NASA makes a chirp and the LED flashes. If the cabin altitude exceeds 12,500 feet, a 30-minute timer starts, and if it stays above 12,500 feet but below 15,000 feet for longer than 30 minutes, an un-mutable, sustained alarm will sound and the LED will flash. This same alarm mode will be in effect if the aircraft cabin pressure exceeds 15,000 feet for any period of time.
SaO2s for 84 passengers not tested for PFO, aged 1–78 years were recorded by pulse oximetry at ground level and during air travel on 10 different airlines at altitudes ranging from 27,000 to 37,000 feet (10). There was a significant reduction in oxygen saturation in all passengers travelling long haul and short haul flights that lasted 1 hour or more. The mean SaO2 for all flights at ground level was 97% [93%–100% +/- 1.33] and at cruising altitude 93% [85%–98% +/-2.33]. Fifty-four per cent of passengers had SaO2 of 94% or less at cruising altitude for which more than one-third of respiratory physicians would prescribe supplementary oxygen (11). No individual had severe cardio-pulmonary problems, and no one required permission from their doctor to fly. Their smoking status was unknown.
Low SaO2 (hypoxia) increases the risk for DVT (Deep Venous Thrombosis) because it increases coagulation, dilates blood vessels and increases capillary permeability. These blood clots in the legs can break off and go to the lungs in a person without a PFO or can go to the brain, heart, or any other organ in a person with a PFO. Other risk factors for causing DVT are reduced blood flow (immobility in the aircraft), dehydration, and damage to blood vessels.
People with a PFO traveling in a Learjet (cabin altitude 8,000-10,000 feet), Boeing 737 (cabin altitude 8,000 feet), or a Boeing 787 (cabin altitude 6,000 feet), will have an % OXYGEN SATURATION lower than 88%-91%, 91%, 93% respectively, and below 95% is considered low.
ALTITUDE DECOMPRESSION SICKNESS
Decompression Sickness results from exposure to low barometric pressures that cause inert gases (mainly nitrogen), normally dissolved in body fluids and tissues, to come out of solution in bubbles and can affect joints, lung, heart, skin and brain. Individual susceptibility can vary from day to day, and different individuals under the same conditions may be affected differently or not at all. It’s just like opening a carbonated soft drink. When the cap is removed from the bottle, the liquid goes from high pressure to a lower barometric pressure, gas is heard escaping, and bubbles can be seen forming in the soda.
Similar to that seen in diving, there is the phenomenon of ALTITUDE DECOMPRESSION SICKNESS in which venous gas emboli can cross a PFO or other potential right to left shunt, move to the arterial side and cause stroke-like symptoms or heart attack. This would require the presence of venous gas emboli and can happen at lower pressure/higher altitude, especially above 18,000 feet. Unfortunately, cases of altitude related neurological Decompression Sickness/Air Gas Emboli are not often evaluated for PFO. In what is an illustrative example (no PFO evaluation done in this case), Dr. Steve Veronneau of the Federal Aviation Administration (FAA) and Dr. Andy Pilmanis of the Air Force Research Laboratory (AFRL) wrote the following short article on a non-fatal case involving a DC-8 in 1994 [NTSB report (12)].
The phenomenon of Altitude Decompression Sickness, whether or not associated with PFO, is well studied in the military literature and has been a particular hazard for pilots operating very high-altitude aircraft, such as the U-2.
RISK FACTORS FOR DEVELOPING ALTITUDE DECOMPRESSION SICKNESS
1) Altitude higher than 18,000 feet
2) Flying an unpressurized aircraft to altitude
3) Fast rate of ascent
4) Mountain climbing
5) Older age
6) Overweight – nitrogen is stored in greater amounts in fat
7) Alcohol
8) Scuba Diving before flying – a person breaths air under high pressure that increases the amount of nitrogen dissolved in the body
SIGNS AND SYMPTOMS OF ALTITUDE DECOMPRESSION SICKNESS
1) BENDS 60%-70% pain mostly in large joints of the body (elbows, shoulders, hip, wrists, knees, ankles). Pain can occur at altitude, during the descent, or many hours later.
2) NEUROLOGIC 10%-15%
BRAIN: Confusion or memory loss • Headache • Spots in visual field (scotoma), tunnel vision, double vision (diplopia), or blurry vision • Unexplained extreme fatigue or behavior changes • Seizures, dizziness, vertigo, nausea, vomiting and unconsciousness may occur
SPINAL CORD • Burning, stinging, tingling around the lower chest and back, abdominal or chest pain • Symptoms may spread from the feet up and may be accompanied by ascending weakness or paralysis.
PERIPHERAL NERVES • Urinary and rectal incontinence • Numbness, burning, stinging and tingling • Muscle weakness or twitching
3) CHOKES Lungs • Burning deep chest pain (under the sternum) • Pain is aggravated by breathing • Shortness of breath • Dry constant cough
4) SKIN BENDS • Itching usually around the ears, face, neck arms, and upper torso • Sensation of tiny insects crawling over the skin
PLATYPNEA-ORTHODEOXIA, a very rare clinical syndrome characterized by shortness of breath and deoxygenation (low oxygen uptake) when a person with a PFO or other atrial septal defect sits or stands from a lying down position thus stretching the hole allowing more streaming of venous blood especially when there is a persistent Eustachian valve. Closure of the hole is the treatment.
CURRENT TREATMENT OF PATIENTS WITH PFO
For many years only the issue of unfiltered blood clots causing a stroke was used to decide how to treat a patient with a PFO because the other important physiological consequences were not considered: (1) Inefficient gas exchange causing hypoxia and carbon dioxide retention, (2) Higher core temperature that also worsens hypoxia, (3) High Altitude effects with or without Altitude Decompression Sickness in a plane or mountain climbing, (4) Unfiltered air bubbles, and (5) Decompression Sickness as in SCUBA diving.
The earlier trials, CLOSURE I (13), RESPECT 2013 (14), and PC Trial (15), showed no difference in outcomes for patients treated by occluding the hole with a mechanical apparatus, or non-surgical management with a blood thinner like coumadin or aspirin. In fact, treatment with aspirin seemed to be best.
The newer trials, CLOSE (16), RESPECT 2017 (17), and REDUCE (18) suggest PFO closure. However, each of these trials had limitations:
(1) there was no consistent dose of aspirin, antiplatelet or anticoagulant within the trials. No trial identified how many people took what aspirin dose (ranged from 75 mg to 325 mg), enteric coated or not, and at what time they took it. Uncoated aspirin 325 mg at 6PM decreases risk for heart attack, stroke, cancer, colon cancer, colon polyps, and decreases lipoprotein(a).
In addition to its antiplatelet effect, 325 mg aspirin inhibits thromboxane A2 much more rapidly than 81 mg and therefore acts more quickly as an anticoagulant.
Aspirin is rapidly absorbed with a bioavailabilty of about 50%, but much lower when given as the enteric coated form. Peak concentration of non-coated aspirin occurs rapidly within 30 minutes after ingestion but up to 4 hours after ingesting enteric coated aspirin (19).
The fibrinolytic system has a circadian rhythm peaking at 6 PM with its lowest level of activity at 6 AM. We are unprotected in the early hours of the morning – in fact, most heart attacks and strokes occur between 3 AM and 5 AM. Therefore, take your non-coated 325 mg aspirin at around 6 PM with your large meal so that you have protection throughout the night.
(2) The REDUCE trial actually showed the same number of silent brain infarcts with or without closure (18).
(3) Complete closure of the PFO in the REDUCE trial was attained by only 75% of patients at the end of 12 months (18).
(4) W.L Gore and Associates, the sponsor of the REDUCE trial, “was responsible for data management and provided the statistician. The sponsor assisted in the creation of the figures and was given the opportunity to review the manuscript.” (18).
Because of these PFO cryptogenic stroke studies, many people may continue aspirin treatment only because: (1) there is a low risk of recurrent stroke with no device; (2) there is doubt of how much benefit the device actually provides; and, (3) there is a risk of complications at the time of device implantation and from having a device in your heart (arrhythmias, clots from the device, etc) [20, 21]. None of these trials, however, considered the important hypoxia physiological consequences described above.
OUR RECOMMENDATION
Mechanically close the hole if the patient has any of the physiological consequences above and there are no medical contraindications for closure.
REFERENCES
1. Hagen PT, Scholz DG, Edwards WD. Incidence and size of patent foramen ovale during the first 10 decades of life: an autopsy study of 965 normal hearts. Mayo Clin Proc. Jan 59(1): 17-20. 1984.
2. Lovering A, Elliott JE, Davis JT. Physiological impact of patent foramen ovale on pulmonary gas exchange, ventilatory acclimatization, and thermoregulation. J Appl Physiol 121: 512–517, 2016.
3. Lovering A, Lozo M, Barak O, Davis JT, Lojpur M, Lozo P, Caljkusic K and Zeljko Dujic. Resting arterial hypoxemia in subjects with chronic heart failure, pulmonary hypertension and patent foramen ovale. Exp Physiol. 101. 5:657-70. 2016.
4. Fenster BE, Nguyen BH, Buckner JK, Freeman AM, Carroll JD. Effectiveness of percutaneous closure of patent foramen ovale for hypoxemia. Am J Cardiol 112: 1258–1262, 2013.
5. Bendz B, Rostrup M, Sevre K, Andersen TO, Sandset PM. Association between acute hypobaric hypoxia and activation of coagulation in human beings. Lancet; 356: 9242; 2000.
6. Majumdar SR, Eurich DT, Gamble JM, Senthilselvan A, Marrie TJ. Oxygen saturations less than 92% are associated with major adverse events in outpatients with pneumonia. Clinical Infectious Diseases, Volume 52(3), 1 February 2011, Pages 325–331.
7. Elliott JE, Laurie SS, Kern JP, Beasley KM, Goodman RD, Kayser B, Subudhi AW, Roach RC, Lovering AT. AltitudeOmics: impaired pulmonary gas exchange efficiency and blunted acclimatization in humans with patent foramen ovale after 16 days at 5,260 m. J Appl Physiol 118: 1100–1112, 2015.
8. Simonson TS, Yang Y, Huff CD, Yun H, Qin G, Witherspoon DJ, Bai Z, Lorenzo FR, Xing J, Jorde LB, Prchal JT, Ge R. Genetic evidence for high-altitude adaptation in Tibet. Science. 329(5987): 72-75. July 2010.
9. Cottrell JJ. Altitude exposures during aircraft flight: flying higher. Chest. 92: 81–4. 1992.
10. Humphreys S, Deyermond R, Bali I, Stevenson M, Fee JP. The effect of high altitude commercial air travel on oxygen saturation. Anesthesia. 2005. 60:458-460.
11. Coker RK, Partridge MR. Assessing the risk of hypoxia in flight: the need for more rational guidelines. European Respiratory Journal. 15: 128–30; 2000.
13. Furlan AJ, Reisman M, Massaro J, Mauri L, Adams H, Albers GW, Felberg R, Herrmann H, et al. Closure or medical therapy for cryptogenic stroke with patent foramen ovale. CLOSURE I. N Engl J Med 2012; 366:991-999March 15, 2012DOI: 10.1056/NEJMoa1009639
14. Carroll JD, Saver JL, Thaler DE, Smalling RW, Berry S, MacDonald LA, Marks DS, Tirschwell DL. Closure of patent foramen ovale versus medical therapy after cryptogenic stroke. RESPECT N Engl J Med 2013; 368:1092-1100 March 21, 2013DOI: 10.1056/NEJMoa1301440
15. Meier B, Kalesan B, Mattle HP, Khattab AA, Hildick-Smith D, Dudek D, Andersen G, Ibrahim R, Schuler G, Walton AS, Wahl A, Windecker S, Jüni P. Percutaneous closure of patent foramen ovale in cryptogenic embolism. PC Trial. N Engl J Med 2013; 368:1083-1091March 21, 2013DOI: 10.1056/NEJMoa1211716
16. Mas JL, Derumeaux G, Guillon B, Massardier E, Hosseini H, Mechtouff L, Arquizan C, et al. Patent foramen ovale closure or anticoagulation vs. antiplatelets after stroke. CLOSE. N Engl J Med 2017; 377:1011-1021September 14, 2017DOI: 10.1056/NEJMoa1705915
17. Saver JL, Carroll JD, Thaler DE, Smalling RW, MacDonald LA, Marks DS, and Tirschwell DL. Long-term outcomes of patent foramen ovale closure or medical therapy after stroke. RESPECT (2017). N Engl J Med 2017; 377:1022-1032September 14, 2017DOI: 10.1056/NEJMoa1610057
18. *Sondergaard, Kasner SE, Rhodes JF, Andersen G, Iversen HK, Nielsen-Kudsk JE, Settergren M, Sjöstrand C, et al. Patent foramen ovale closure or antiplatelet therapy for cryptogenic stroke. REDUCE, N Engl J Med 2017; 377:1033-1042September 14, 2017DOI: 10.1056/NEJMoa1707404.
* All authors report Personal Fees, and/or Grants, and/or “Other” from W.L. Gore and Associates that funded the study.
19. Patrono C. Aspirin. Chapter 53. Platelets. Academic Press. 2013. Pages 1099-1115.
20. Abaci A, Unlu S, Alsancak Y, Sezenoz B. Short and long term complications of device closure of atrial septal defect and patent foramen ovale: Meta-analysis of 28,142 patients from 203 studies. Cathet Cardiovasc Intervent. 82: 1123–1138. 2013.
21. Mirzada N, Ladenvall P, Hansson PO. Seven-year follow-up of percutaneous closure of patent foramen ovale. IJC Heart and Vasculature. 1:32-36. 2013.
(c) 2017 Charles B. Simone, M.MS., M.D.